Professional Perspectives: Adverse Outcome Pathways to Contextualize Mechanistic Data for Chemical and Radiation Research
By Vinita Chauhan | April 9th, 2026
Risk assessment in both chemical and radiation research has long relied on decades of evidence from animal studies and human epidemiology. At the same time, advances in cell-based systems, computational modeling, and molecular biology are generating new forms of mechanistic information. The challenge now lies in how to systematically interpret and contextualize these data within established risk frameworks.
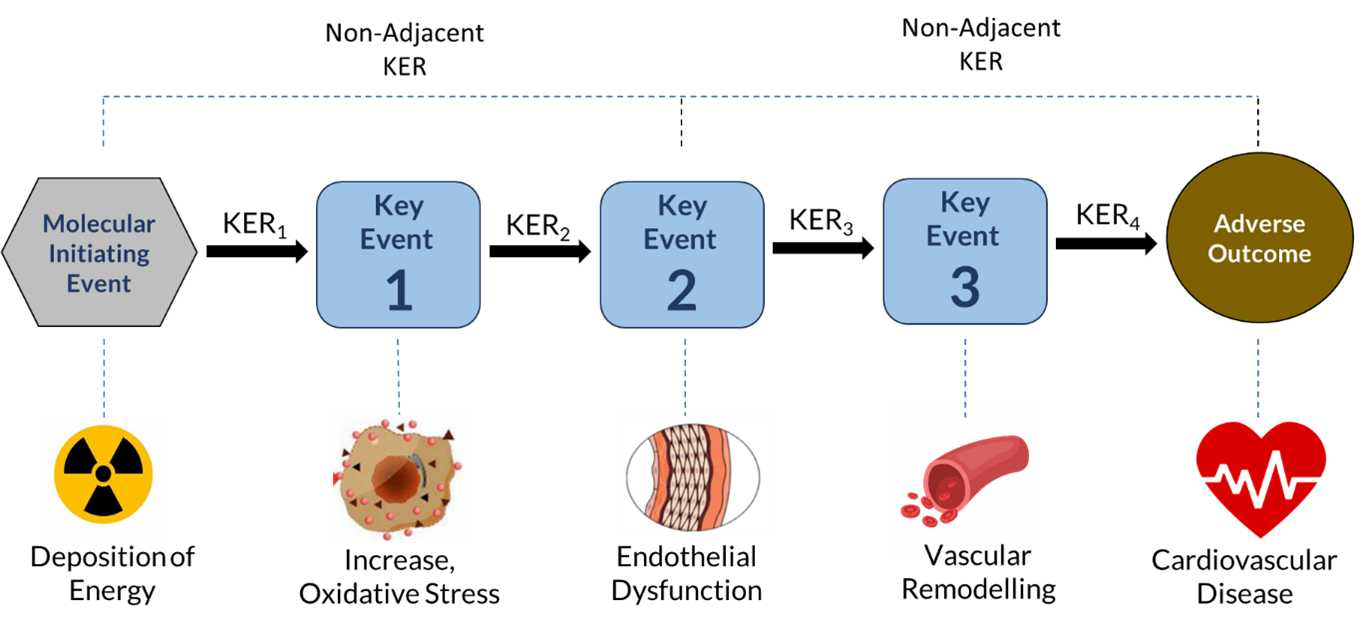
Adverse Outcome Pathways (AOPs) provide one structured approach for organizing such information. An AOP describes a sequence of causally linked biological events, beginning with a molecular initiating event and progressing through measurable key events to an adverse outcome (Building AOPs). By mapping biological processes across levels of organization, AOPs identify the relationships between early molecular perturbations and later functional or clinical effects.
Although originally developed in chemical toxicology, the AOP framework is increasingly being explored within radiation biology. Collaborative efforts under the Organisation for Economic Cooperation and Development (OECD) Nuclear Energy Agency (NEA) AOP Radiation/Chemical (Rad/Chem) Joint Topical Group ( OECD NEA RAD/CHEM AOP Joint Topical Group ) reflect recognition that many biological responses including DNA damage, oxidative stress, and inflammatory pathways are shared across chemical and radiological stressors.
Several radiation-relevant examples (Deposition of Energy AOPs; Non Cancer AOP video) illustrate this potential. Deposition of ionizing radiation induces DNA strand breaks and activates damage response pathways. These early molecular events may trigger downstream processes such as persistent oxidative stress or inflammatory signaling. In some tissues, sustained activation of these pathways has been associated with structural and functional changes. Framing this sequence within an AOP clarifies which linkages are well supported, which remain uncertain, and where additional data may be required.
Advanced experimental and computational approaches now generate more detailed mechanistic evidence. Within the AOP framework, this new information can be organized, interpreted, and weighted alongside traditional endpoints. AOPs help researchers distinguish between well-supported biological pathways and emerging hypotheses, guiding research planning. This is particularly valuable in areas of high uncertainty, such as low-dose exposures (e.g., medical diagnostic procedures like CT scans and environmental sources such as radon exposure in homes), non-targeted effects (e.g., bystander effects and adaptive responses), and variability in individual susceptibility. Importantly, AOPs do not replace established lines of evidence. Epidemiological observations and in vivo studies remain central to risk assessment. Rather, structured incorporation of mechanistic data can improve transparency in how biological plausibility and causal coherence are evaluated alongside traditional endpoints.
In this sense, applying AOPs should therefore be viewed as a methodological refinement rather than a paradigm shift. It offers a systematic way to assemble mechanistic information, identify data gaps, and communicate uncertainty more clearly. As emerging science continues to expand, such structured approaches may support more consistent interpretation of evidence across both chemical and radiation research domains.
Dr. Vinita Chauhan is a Senior Research Scientist with the Consumer and Clinical Radiation Protection Bureau (CCRPB) at Health Canada, with over 20 years of experience in radiobiology. She earned her Ph.D. in Biochemistry from the University of Ottawa Heart Institute in 2000, where her research focused on lipid composition and low density lipoprotein receptor binding. She later completed a postdoctoral fellowship at Health Canada, working on environmental pollutants and in vitro toxicity assays before transitioning to CCRPB in 2005 to study the effects of non-ionizing radiation on brain cells using transcriptional technologies. Currently, she works in the Ionizing Health Sciences Division, focusing on low-dose radiation research through biomarker identification using multi-omics and adverse outcome pathways (AOPs).
Dr. Chauhan has published over 90 scientific papers and received the Health Canada Assistant Deputy Minister Award in 2023 in recognition of her contributions to AOPs. She plays an active leadership role internationally as a nominated delegate to the OECD Emerging Science in Chemicals Assessment Advisory Group and co-chair of the Rad/Chem AOP Joint Topical Group within the Nuclear Energy Agency High-Level Group on Low Dose. She also co-coordinates the Canadian Organization of Health Effects from Radiation Exposure (COHERE) initiative with the Canadian Nuclear Safety Commission, working to advance national coordination of low-dose radiation research in Canada.
The views expressed do not necessarily reflect the official policy or position of Johns Hopkins University or Johns Hopkins Bloomberg School of Public Health.