Registration is closed! Please contact Loza Taye (ltaye1@jh.edu) for updates on the workshop report.
A two-day hybrid workshop will be hosted by the Johns Hopkins Toxicology Policy Lab and Cruelty Free International to explore the ICH M3 (R2) guidance on second species testing in light of the most current scientific evidence. Participants will critically examine the current requirements for two-species toxicity testing, including whether available evidence supports greater flexibility in the requirement for a second species in long-term studies.
Featured Speakers:
C. Lee Elmore, US Food and Drug Administration (FDA)
Sonja Beken, Federal Agency for Medicines and Health Products (Belgium); EMA 3RsWP
Fiona Sewell, National Centre for the Replacement, Refinement, and Reduction of Animals in Research (NC3Rs)
Frank Bringezu, Merck Healthcare KGaA
Steering Committee:
Warren Casey, National Institutes of Health (NIH)
Thomas Hartung, Johns Hopkins Center for Alternatives to Animal Testing (CAAT)
Sally Thompson-Iritani, University of Washington; 3Rs Collaborative
Szczepan Baran, Instem; Baran Café
Nathalie Printemps, Corteva; European Partnership for Alternative Approaches to Animal Testing (EPAA)
Paul Locke, Johns Hopkins Toxicology Policy Lab
Context:
The recent revision of the ICH S1 guideline introduced a WoE approach that allows the two-year rodent carcinogenicity study to be waived under certain conditions. This change followed a comprehensive review of the study’s scientific value, setting an important precedent for evidence-based updates to longstanding regulatory testing requirements. In contrast, the ICH M3 guideline, which mandates toxicity testing in two animal species for small molecule pharmaceuticals, has not been updated in many years despite advances in science and increasing public and political pressure to reduce animal testing. Notably, there is growing concern about testing on dogs and non-human primates, species often used as the second test species. These pressures are reflected in global strategies aimed at phasing out animal tests, including the FDA’s most recent roadmap to reduce animal testing and the goals outlined in the FDA Modernization Act. This workshop will examine the scientific rationale for routinely conducting long-term toxicity studies in two species under ICH M3(R2), but in decision terms rather than as a binary retention-or-removal question. It should ask what specific biological question remains unresolved after earlier two-species studies, ADME and metabolite characterization, pharmacologic relevance assessment, exposure-margin analysis, safety pharmacology, and available fit-for-purpose NAMs. Where value is context-dependent, the discussion should define the evidence standards that permit bounded flexibility.
Session 1: Lessons from ICH S1
Designing a robust review - what a credible, regulator-led evaluation must include
What worked in S1’s prospective evaluation
What challenges emerged (subjectivity, regulator/sponsor differences, data gaps)
What an M3 review might need to evaluate the two-species paradigm
Pros/cons of prospective vs. retrospective approaches in the M3 context
Session 2: Evidence Needs & Evaluation Pathways
Identify data needs
Types of analyses ICH should conduct using proprietary data
Criteria for assessing when the second species adds meaningful value
How to incorporate mechanistic understanding, relevance, and NAMs
Key scientific questions that an ICH M3 working group should address
Session 3: Regulatory Options & Practical Pathways for Flexibility
Identify actionable outputs for ICH
Operational considerations (global alignment, review processes)
Drafting key elements of a “blueprint” for an ICH-led review
Consensus on priority questions and study design recommendations
Deliverables:
A published workshop report outlining study design(s), metrics, and data needs for a potential ICH review of M3.
An evidence-based options list for reducing or refining the two- species requirement while maintaining human safety.
A roadmap for next steps, including pilot studies and data-sharing mechanisms.
Date and Location
The two-day workshop will be held in person at Johns Hopkins University Bloomberg Center in Washington DC or online on the following dates:
Day 1: May 21st, 2026: 9:00AM - 4:30PM EST
Day 2: May 22nd, 2026: 9:00AM - 12:30PM EST
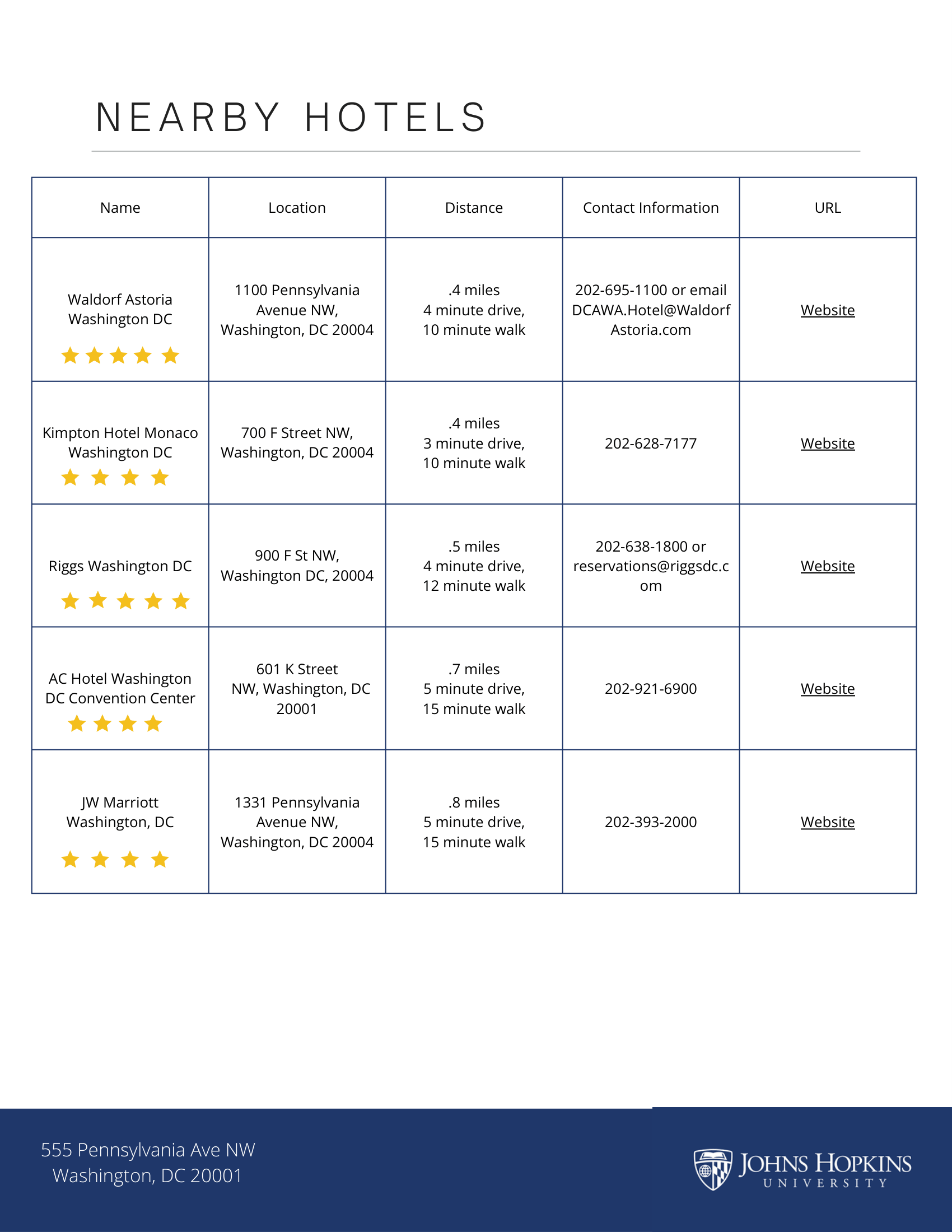
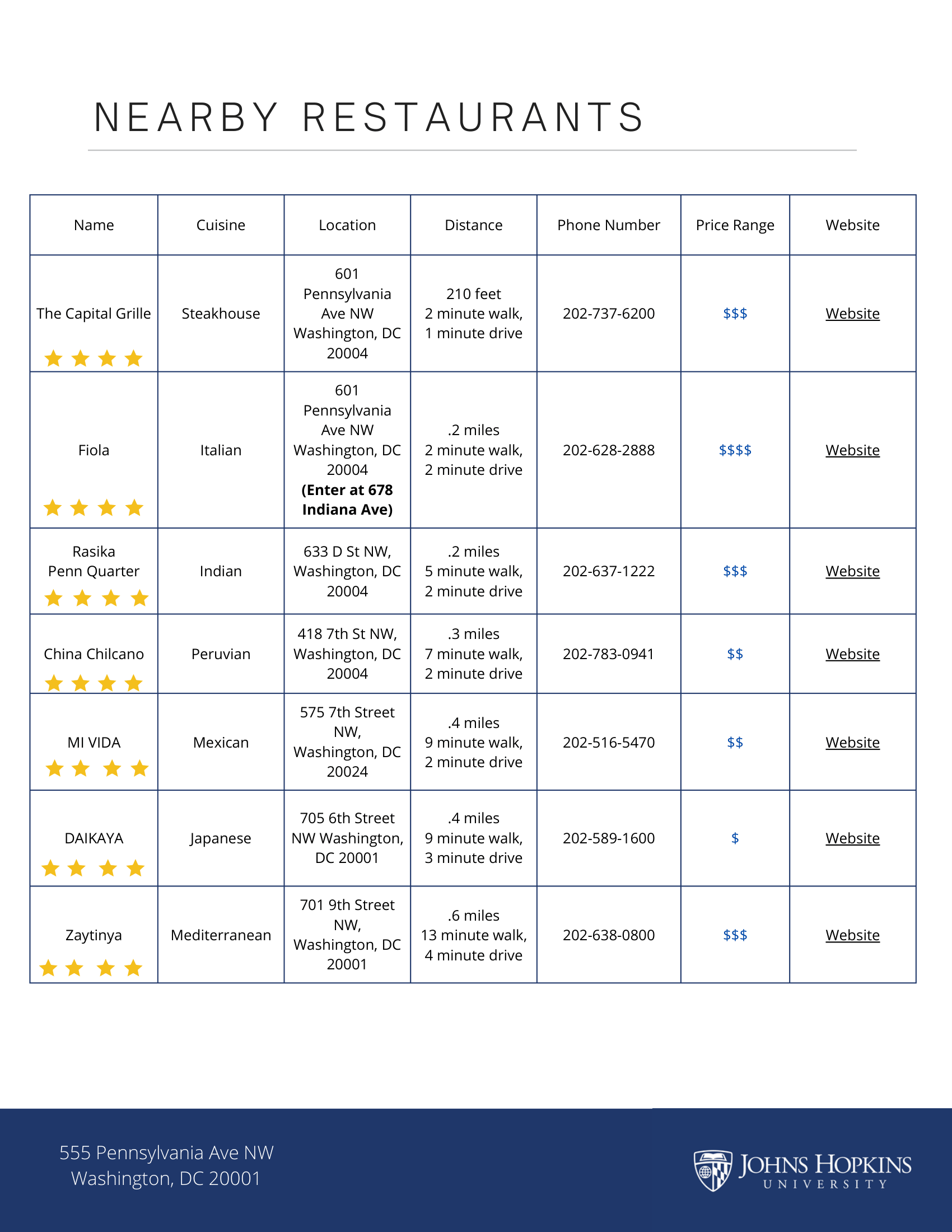
In-Person Location: 555 Pennsylvania Ave NW, Washington, DC 20001, Room 1024
Agenda
Resources:
To prepare for the workshop, you may find these documents helpful:
Food and Drug Administration, HHS. International Conference on Harmonisation; Guidance on M3(R2) Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals; availability. Notice. Fed Regist. 2010 Jan 21;75(13):3471-2. PMID: 20349552.
NC3Rs. Review of the use of two species in regulatory toxicology studies (ICH M3). National Centre for the Replacement, Refinement and Reduction of Animals in Research. Available from: https://nc3rs.org.uk/our-portfolio/review-use-two-species-regulatory-toxicology-studies-ichm3
Bourcier T, McGovern T, Cavaliero T, Ebere G, Nishikawa A, Nishimura J, Ogawa K, Pasanen M, Vespa A, Van der Laan JW. ICH S1 prospective evaluation study: weight of evidence approach to predict outcome and value of 2-year rat carcinogenicity studies. A report from the regulatory authorities subgroup. Front Toxicol. 2024 Apr 11;6:1353783. doi: 10.3389/ftox.2024.1353783. PMID: 38665214; PMCID: PMC11043531
Bailey J, Thew M, Balls M. An analysis of the use of dogs in predicting human toxicology and drug safety. Altern Lab Anim. 2013 Nov;41(5):335-50. doi: 10.1177/026119291304100504. PMID: 24329742.
Bailey J, Thew M, Balls M. Predicting human drug toxicity and safety via animal tests: can any one species predict drug toxicity in any other, and do monkeys help? Altern Lab Anim. 2015 Dec;43(6):393-403. doi: 10.1177/026119291504300607. PMID: 26753942.
Prior H, Baldrick P, de Haan L, Downes N, Jones K, Mortimer- Cassen E, Kimber I. Reviewing the Utility of Two Species in General Toxicology Related to Drug Development. Int J Toxicol. 2018 Mar;37(2):121–4. doi: 10.1177/1091581818760564. Epub 2018 Mar 27. PMCID: PMC5881785.